A unified platform that organizes complex FDA datasets, drugs, devices, approvals, and recalls—into fast, accurate, and easy-to-understand insights.

Turning scattered FDA records into one reliable source of truth.
CORE Approach
FDA information is vast, fragmented, and inconsistent. Critical details are buried across APIs, PDFs, and decades of device and drug listings.
We built a centralized intelligence layer that cleans, aligns, and enriches these datasets—making it possible to explore approvals, timelines, recalls, and classifications with clarity.
The Core Intent of Our Approach
The goals that defined our direction, ensuring every
signal becomes clearer, smarter, and more meaningful.
Unify Data Sources
All FDA APIs, PDFs, device summaries, and drug listings are consolidated into a single, stable system that eliminates fragmentation and data loss.
Transform raw records
Every dataset goes through noise removal, formatting alignment, enrichment layers, and schema stabilization—ensuring high-quality, trustworthy results.
Preserve Critical Insights
Important regulatory metadata—approval dates, product codes, classifications, summaries—is extracted and retained with full traceability to original sources.
Build Intelligent Access
A search layer that understands product names, companies, codes, and timelines, offering fast retrieval and meaningful filtering across all regulatory categories.
THE PROBLEM
Before clarity was possible, each signal arrived with layers of distortion, drift, and unpredictable behavior that made reliable interpretation nearly impossible.
Inconsistent records
Different endpoints return missing, outdated, or unstructured fields.
Fragmented sources
Electrical interference, muscle activity, and sensor vibration overwhelming key features.
Limited Visibility
Historical patterns and timelines are hard to compare manually.
Complex metadata
Codes and classifications rarely align across systems.
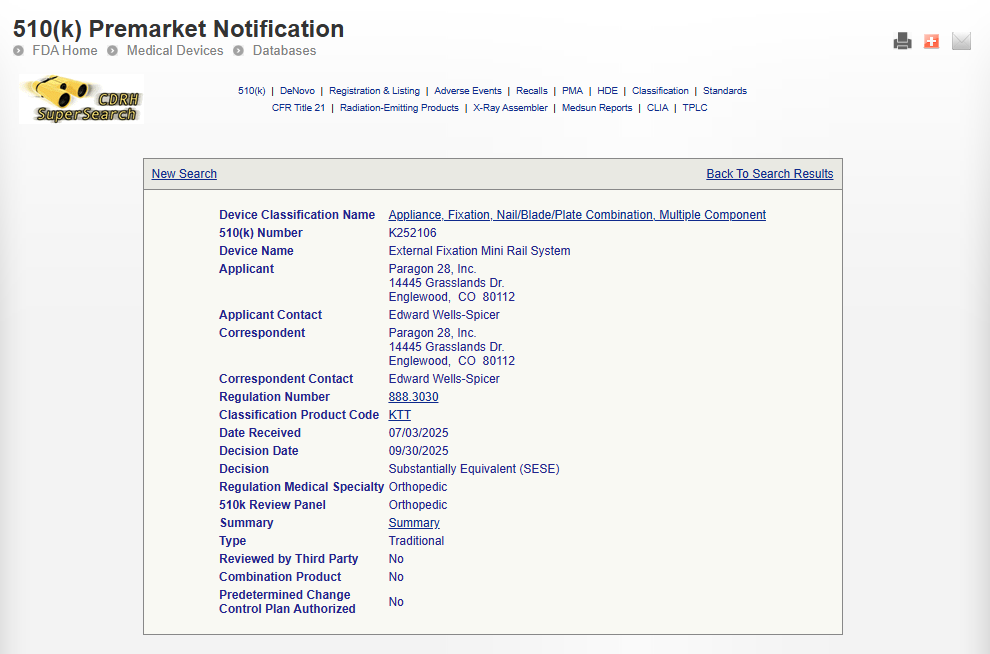
WORKFLOW
We streamlined every dataset into a stable, structured, and easy-to-explore system.
Nightly sync pipelines fetch, validate, and update all FDA datasets reliably.
Normalize
every record
Formats, codes, dates, and identifiers are cleaned and standardized across sources.
Extract
key insights
Approvals, classifications, summaries, recalls, and timelines are isolated with accuracy.
Enable
smart access
Fast search and filters make regulatory exploration simple and reliable.
TECH INSIGHTS
Engineering the core behind
FDA intelligence
Key decisions that shaped a fast, accurate, and scalable regulatory platform.
AI
Extraction
Modular
APIs
Instant
Alerts
AWS
Delivery
Cloud infrastructure ensures secure, stable, and globally optimized access.

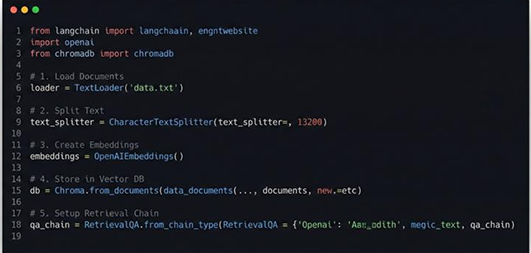
Result
With noise removed and patterns restored, each signal became a trustworthy foundation for analysis, monitoring, and intelligent
decision-making.
Accurate Regulatory Insights
|
Unified FDA Data Access
|
Reliable Approval Tracking
|
Search-Ready Structured Records
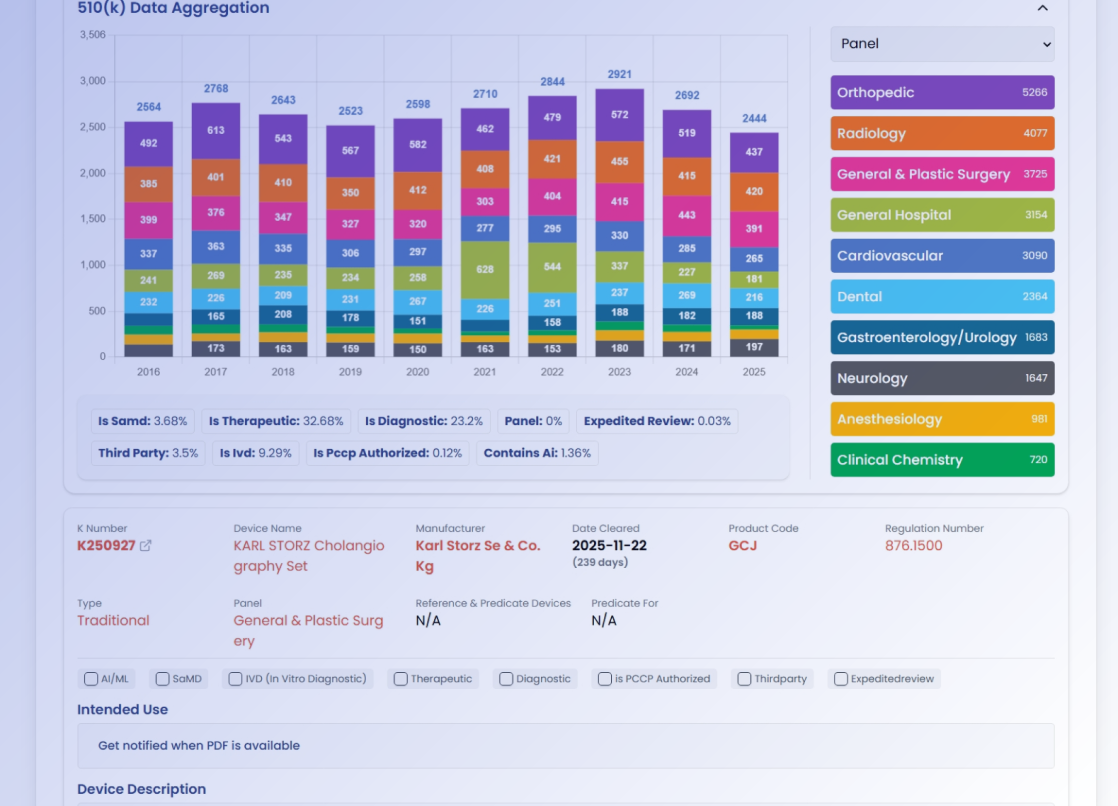
CASE STUDIES
Explore related projects with similar challenges and outcomes.